Intracellular phase transitions
The conventional view of intracellular organization is focused on the cell’s use of membrane-bound, vesicle-like organelles to compartmentalize the cytoplasm. However, in addition to these canonical organelles, there exist many membrane-less organelles within living cells. These membrane-less structures are referred to as intracellular condensates or simply condensates. These include ribonucleoprotein (RNP) condensates such as processing bodies and stress granules in the cytoplasm, and Cajal bodies and nucleoli in the nucleus.
Many intracellular condensates behave in a manner consistent with phase-separated liquids. Such behaviors include: (1) They typically exhibit round morphologies; (2) they readily fuse with one another on contact; (3) they exhibit rapid exchange of component molecules with surrounding nucleoplasm or cytoplasm; (4) they can exhibit wetting behavior, e.g., P granules exhibit a well-defined contact angle on the nuclear envelope; (5) they can exhibit these dynamic fluid properties, and yet do not mix with one another, much like two immiscible liquid phases; and (6) they only assemble above a threshold concentration of their components.
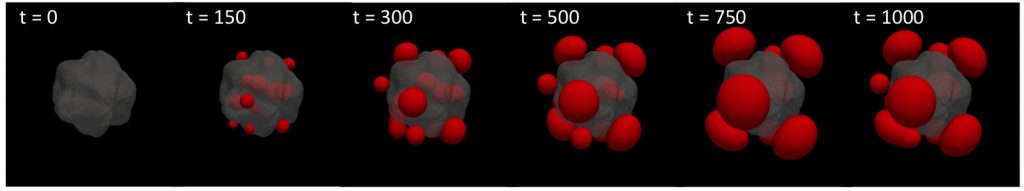
We are currently investigating such phase separation processes in living systems via a combination of analytical and computational tools. For example, many intrinsically disordered proteins phase separate into liquid droplets and interact with chromatin. The animation at the top of the page and figure below display diffuse-interface simulations of a mechanical droplet exclusion model with activation localized within a stiff heterochromatin-like domain (gray contour). Following nucleation of small droplets within the activated region, mechanical deformation energy drives droplets and droplet-forming molecules to the exterior of the heterochromatin-like domain where large condensates ultimately accumulate and coarsen.
More information can be found in J. Berry et al., PNAS (2015), Y. Shin et al., Cell (2017), J. Berry et al., ROPP (2018), and Y. Shin et al., Cell (2018).